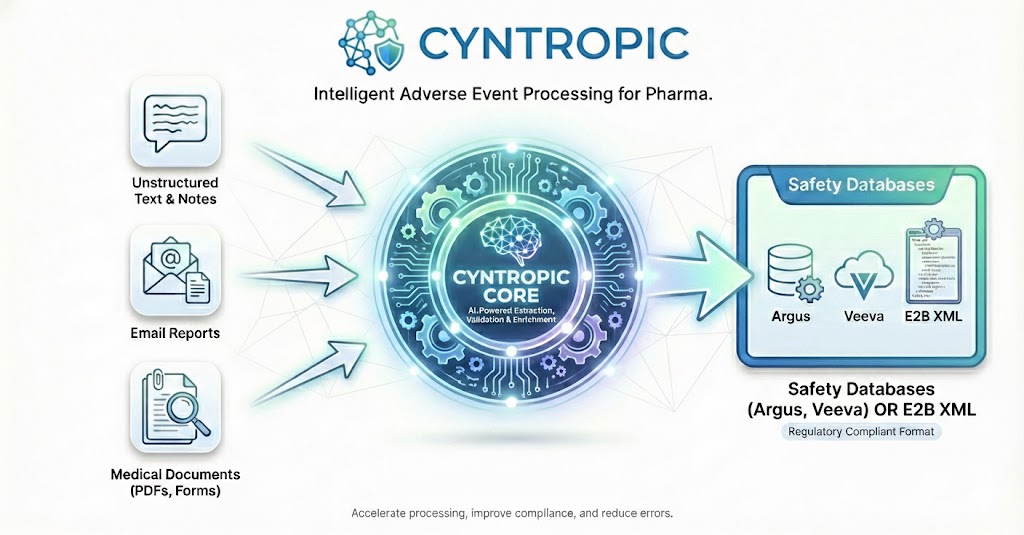
Next-Generation Pharmacovigilance
AI-powered with a risk-based approach prioritizing human oversight
Cyntropic streamlines the safety lifecycle with proprietary, compliant AI - from intake to submission - to turn your safety operations into a transparent, auditable, and cost-efficient asset.